
瑕瑜课题组 | 施恒学的博士生涯
2026-02-04
大家好,我是清华大学化学系瑕瑜课题组 2019 级博士生施恒学,于2025年6月获得博士学位。应瑕老师要求,回首博士生涯,很荣幸与大家分享我的成长点滴。我本科就读兰州大学化学专业,当时对科研并无清晰认知,也难以确定自己是否怀有科研热忱。2018年,抱
[ [{ "name": 1, "label": "日本料理" }, { "name": 2, "label": "韩式料理" }, { "name": 3, "label": "意大利料理" }, { "name": 4, "label": "法国料理" }, { "name": 5, "label": "中东菜" }, { "name": 6, "label": "东南亚菜" }], [{ "name": 7, "label": "北京菜" }, { "name": 8, "label": "上海菜" }, { "name": 9, "label": "江浙菜" }, { "name": 10, "label": "粤菜" }, { "name": 11, "label": "闽菜" }, { "name": 12, "label": "湘菜" }, { "name": 13, "label": "徽菜" }, { "name": 14, "label": "苏菜" }, { "name": 15, "label": "鲁菜" }, { "name": 16, "label": "川渝火锅" }, { "name": 17, "label": "云贵菜" }, { "name": 18, "label": "江西菜" }, { "name": 19, "label": "其他菜系" }] ]
探索我们的研究
2026-02-04
大家好,我是清华大学化学系瑕瑜课题组 2019 级博士生施恒学,于2025年6月获得博士学位。应瑕老师要求,回首博士生涯,很荣幸与大家分享我的成长点滴。我本科就读兰州大学化学专业,当时对科研并无清晰认知,也难以确定自己是否怀有科研热忱。2018年,抱
2026-02-04
王紫丹 博士瑕瑜课题组感谢贺思敏老师的邀请和瑕瑜老师的推荐,让我有机会回顾这段充满挑战与惊喜的博士生涯。我本科就读于安徽大学应用化学专业。2017年,我怀揣着对科研的热忱,踏入复旦大学,师从邓春晖教授,基于功能性磁性纳米材料的设计,开发翻译后修饰蛋白
2026-02-05
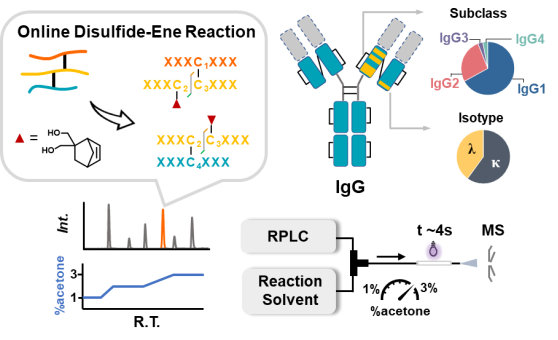
近期,清华大学化学系瑕瑜课题组开发了一种在线二硫键-烯反应平台,通过在线调节反应溶剂的组成,在液相色谱分离后的二硫键肽段中实现最佳的部分还原效果,从而实现高覆盖率的二硫键鉴定。该项研究“High-coverage disulfide mapping
2026-02-05
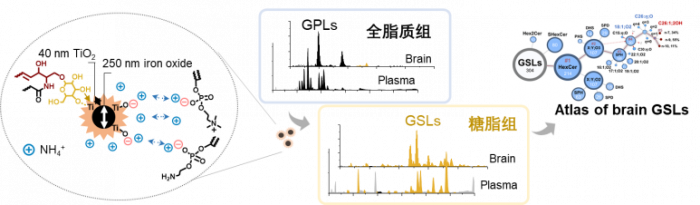
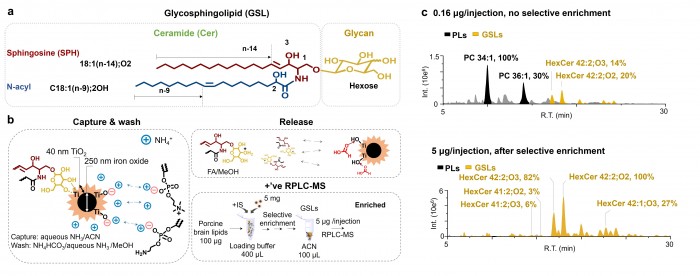
中性鞘糖脂是一种重要的两亲性脂质分子,其在细胞膜运输和信号传导过程中发挥作用。此外,中性鞘糖脂分子水平上的变化与多种神经疾病、代谢紊乱和癌症相关。由于鞘糖脂丰度低、离子化效率低、结构复杂,已有的分析方法对中性鞘糖脂组的鉴定覆盖率一般低于20%。近期清
2026-02-05
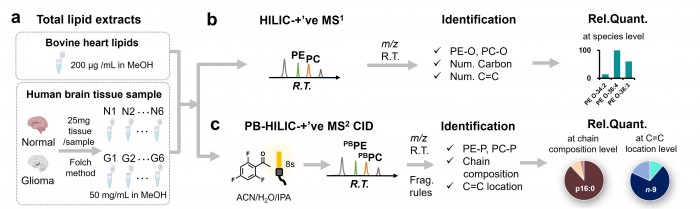
近期,清华大学化学系瑕瑜教授课题组在Analytical and Bioanalytical Chemistry杂志上发表了题为 “Deep profiling of plasmalogens by coupling the Paternò–Büch
AbstractPlants can adapt to environmental fluctuations through modulating their fatty acids (FAs) dynamically. In this study, an enhanced mass spectrometry approach is utilized to uncover an unexplored landscape of FAs and FA isomers that are critical for cold tolerance in tomato. This technology integrates N‐(4‐aminomethylphenyl) pyridium derivatization of FAs, charge‐tagging Paternò‐Büchi (PB) photochemical reaction to identify carbon–carbon double bond (C═C) positions and reversed‐phase liquid chromatography coupled with tandem mass spectrometry to achieve efficient detection of FAs and their C═C location isomers. Several saturated FAs, unsaturated FAs and their C═C location isomers are revealed to contribute to the cold tolerance of elongated hypocotyl 5 (slhy5) and fatty acid desaturase (slfad) mutant plants. RNA‐sequencing analysis and dual‐luciferase reporter assays further demonstrate that SlHY5 can modulate the expression of SlFAD2 genes under cold stress, regulating FA desaturation. The application of FA isomers to the leaves of slfad mutants partially rescues their cold sensitivity, presenting the practical implications of the study. The study thereby highlights the importance of considering isomeric variations in FAs when investigating plant physiology and stress responses. Furthermore, this methodology sets a valuable precedent for future investigations aimed at unraveling the intricate metabolic networks that govern plant stress adaptation.
AbstractAlthough GPR3 plays pivotal roles in both the nervous system and metabolic processes, such as cold-induced thermogenesis, its endogenous ligand remains elusive. Here, by combining structural approach (including cryo-electron microscopy), mass spectrometry analysis, and functional studies, we identify oleic acid (OA) as an endogenous ligand of GPR3. Our study reveals a hydrophobic tunnel within GPR3 that connects the extracellular side of the receptor to the middle of plasma membrane, enabling fatty acids to readily engage the receptor. Functional studies demonstrate that OA triggers downstream Gs signaling, whereas lysophospholipids fail to activate the receptor. Moreover, our research reveals that cold stimulation induces the secretion of OA in mice, subsequently activating Gs/cAMP/PKA signaling in brown adipose tissue. Notably, brown adipose tissues from Gpr3 knockout mice do not respond to OA during cold stimulation, reinforcing the significance of GPR3 in this process. Finally, we propose a “born to be activated and cold to enhance” model for GPR3 activation. Our study provides a starting framework for the understanding of GPR3 signaling in cold-stimulated thermogenesis.
AbstractGlycosphingolipids (GSLs) are essential components of cell membranes, particularly enriched in the nervous system. Altered molecular distributions of GSLs are increasingly associated with human diseases, emphasizing the significance of lipidomic profiling. Traditional GSL analysis methods are hampered by matrix effect from phospholipids and the difficulty in distinguishing structural isomers. Herein, we introduce a highly sensitive workflow that harnesses magnetic TiO2 nanoparticle-based selective enrichment, charge-tagging Paternò–Büchi reaction, and liquid chromatography-tandem mass spectrometry. This approach enables mapping over 300 distinct GSLs in brain tissues by defining sugar types, long chain bases, N-acyl chains, and the locations of desaturation and hydroxylation. Relative quantitation of GSLs across multiple structural levels provides evidence of dysregulated gene and protein expressions of FA2H and CerS2 in human glioma tissue. Based on the structural features of GSLs, our method accurately differentiates human glioma with/without isocitrate dehydrogenase genetic mutation, and normal brain tissue.
AbstractBackgroundAdaptative desaturation in fatty acid (FA) is an emerging hallmark of cancer metabolic plasticity. Desaturases such as stearoyl‐CoA desaturase (SCD) and fatty acid desaturase 2 (FADS2) have been implicated in multiple cancers, and their dominant and compensatory effects have recently been highlighted. However, how tumors initiate and sustain their self‐sufficient FA desaturation to maintain phenotypic transition remains elusive. This study aimed to explore the molecular orchestration of SCD and FADS2 and their specific reprogramming mechanisms in response to cancer progression.MethodsThe potential interactions between SCD and FADS2 were explored by bioinformatics analyses across multiple cancer cohorts, which guided subsequent functional and mechanistic investigations. The expression levels of desaturases were investigated with online datasets and validated in both cancer tissues and cell lines. Specific desaturation activities were characterized through various isomer‐resolved lipidomics methods and sensitivity assays using desaturase inhibitors. In‐situ lipid profiling was conducted using multiplex stimulated Raman scattering imaging. Functional assays were performed both in vitro and in vivo, with RNA‐sequencing employed for the mechanism verification.ResultsAfter integration of the RNA‐protein‐metabolite levels, the data revealed that a reprogramming from SCD‐dependent to FADS2‐dependent desaturation was linked to cancer epithelial‐mesenchymal transition (EMT) and progression in both patients and cell lines. FADS2 overexpression and SCD suppression concurrently maintained EMT plasticity. A FADS2/β‐catenin self‐reinforcing feedback loop facilitated the degree of lipid unsaturation, membrane fluidity, metastatic potential and EMT signaling. Moreover, SCD inhibition triggered a lethal apoptosis but boosted survival plasticity by inducing EMT and enhancing FA uptake via adenosine monophosphate‐activated protein kinase activation. Notably, this desaturation reprogramming increased transforming growth factor‐β2, effectively sustaining aggressive phenotypes and metabolic plasticity during EMT.ConclusionsThese findings revealed a metabolic reprogramming from SCD‐dependent to FADS2‐dependent desaturation during cancer EMT and progression, which concurrently supports EMT plasticity. Targeting desaturation reprogramming represents a potential vulnerability for cancer metabolic therapy.
AbstractOxidation reactions are fundamental transformations in organic synthesis and chemical industry. With oxygen or air as terminal oxidant, aerobic oxidation catalysis provides the most sustainable and economic oxidation processes. Most aerobic oxidation catalysis employs redox metal as its active center. While nature provides non-redox metal strategy as in pyrroloquinoline quinone (PQQ)-dependent methanol dehydrogenases (MDH), such an effective chemical version is unknown. Inspired by the recently discovered rare earth metal-dependent enzyme Ln-MDH, here we show that an open-shell semi-quinone anionic radical species in complexing with lanthanum could serve as a very efficient aerobic oxidation catalyst under ambient conditions. In this catalyst, the lanthanum(III) ion serves only as a Lewis acid promoter and the redox process occurs exclusively on the semiquinone ligand. The catalysis is initiated by 1e--reduction of lanthanum-activated ortho-quinone to a semiquinone-lanthanum complex La(SQ-.)2, which undergoes a coupled O-H/C-H (PCHT: proton coupled hydride transfer) dehydrogenation for aerobic oxidation of alcohols with up to 330 h−1 TOF.
By coupling O-benzylhydroxylamine derivatization and tandem mass spectrometry, nitroxide radical-induced dissociation can be initiated via collisional activation which enables the analysis of methyl branching(s) in fatty acids.